
One of the most critical factors limiting the large- scale offtake and deployment of green hydrogen is its cost. The high power consumption required for water electrolysis (≥50 kWh/kg H2 ) remains a major challenge. Despite ongoing technological advancements, achieving the desired green hydrogen price target of ≤$2/kg H2 through electrolysis remains difficult. Approximately 60% of the operating cost of an electrolyser plant arises from renewable electricity consumption. Therefore, reducing electricity demand is essential.
It is well known that the oxidation of many organic molecules, such as glucose, glycerol and alcohols, occurs at significantly lower potentials. This raises an important question: can hydrogen generation at the cathode be integrated with the oxidation of organic molecules at the anode to produce value- added products while operating the electrolyser at a lower potential?
This approach offers a promising pathway to produce low-cost hydrogen. By operating the electrolyser at lower potentials than conventional water electrolysis, organic or biomass- derived molecules can be oxidised to valuable products at the anode (please refer ACS Applied Materials Interfaces 16, 26130, 2024; Chemical Engineering Journal 529, #172633, 2026). In conventional electrolysis, oxygen of relatively low value is produced at the anode and requires a high potential and significant electricity input. In contrast, the proposed method simultaneously reduces the required voltage and generates value-added chemicals.
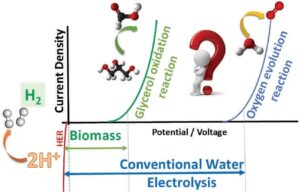
Figure 1: Schematic display to show the voltage reduction with organic/biomass-based electrolysis, compared to conventional water electrolysis
A schematic representation of voltage reduction alongside the formation of value-added products is shown in Figure 1.
It should be noted that the molecular weight of organic products formed at the anode is much higher than that of hydrogen (H2 ). For example, the conversion of methanol (CH3 OH; 32 g) to formaldehyde (28 g) while producing green hydrogen (H2 ; 2 g) requires the same number of charges at the anode and cathode. Consequently, for every kg of hydrogen produced, approximately 14 kg of acetaldehyde could also be generated. By comparison, conventional electrolysis co- produces around 9 kg of oxygen (O2 ) per kg of hydrogen, which has relatively low economic value.
Methanol is a small molecule, but larger molecules such as glycerol and glucose can also be oxidised. As the molecular weight of the feedstock increases, the mass of the product increases proportionally while the hydrogen yield remains fixed at 2 g per reaction unit. Importantly, the entire process operates at significantly lower electricity input. Moreover, the operational cost of the electrolyser plant can be distributed between hydrogen production and anodic value-added products, depending on the nature and market value of the product.
These findings raise the possibility of converting even larger molecules, such as cellulose or certain polymers, into valuable products. However, research on such heavy molecules remains limited for several technical reasons. Greater progress has been achieved with molecules such as glycerol, a by-product of biodiesel production. Glucose, for instance, is a fundamental building block of biomass structures such as cellulose.
At a broader level, biomass-derived molecules could be converted into high-value chemicals. For example, glucose can be oxidised to glucaric acid, an important pharmaceutical intermediate. Similarly, polymers such as polyethylene could potentially be converted into long-chain fuel molecules
including petrol (gasoline) and diesel.
This integration links green hydrogen production with the chemical, pharmaceutical and petrochemical sectors, all of which can utilise both hydrogen and the co-produced chemicals. Such integration could significantly alleviate the current offtake challenge for green hydrogen.
An even broader implication is the recycling of polymers and biomass components within a carbon-neutral economy. Bio- based electrolysis not only produces valuable fuels and chemicals but also helps mitigate environmental pollution through material utilisation and recycling. In addition, value- added product formation typically occurs through stepwise reactions with fast kinetics, enabling higher product formation rates compared with conventional electrolysis.
Despite these advantages, challenges remain. Oxidation of organic molecules often produces multiple products at the anode. Therefore, the anode catalyst and operating conditions must be carefully optimised to achieve selective production of the desired product. Even with high selectivity, additional product separation steps may be required.
These aspects require further evaluation. It is therefore essential for the scientific community to intensify research efforts and develop core technologies in this emerging sector.
Insights shared by: Prof. Chinnakonda S. Gopinath, Visiting Professor, Department of Chemistry, IIT Palakkad
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter, Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!













